
-
 'Unsustainable' housing crisis bedevils Spain's socialist govt
'Unsustainable' housing crisis bedevils Spain's socialist govt
-
Stocks shrug off China disappointment but oil slides

-
 Stocks diverge, oil retreats as China disappoints markets
Stocks diverge, oil retreats as China disappoints markets
-
Trio wins economics Nobel for work on wealth inequality

-
 Ex-Stasi officer jailed over 1974 Berlin border killing
Ex-Stasi officer jailed over 1974 Berlin border killing
-
Shanghai stocks gain after stimulus briefing as markets rally

-
 Shanghai stocks gain after stimulus briefing as Asian markets rally
Shanghai stocks gain after stimulus briefing as Asian markets rally
-
Nearly 90, but opera legend Kabaivanska is still calling tune

-
 With inflation down, ECB eyes faster tempo of rate cuts
With inflation down, ECB eyes faster tempo of rate cuts
-
Is life possible on a Jupiter moon? NASA goes to investigate

-
 Ex-Stasi officer faces verdict over 1974 Berlin border killing
Ex-Stasi officer faces verdict over 1974 Berlin border killing
-
Role of government, poverty research tipped for economics Nobel

-
 In milestone, SpaceX 'catches' megarocket booster after test flight
In milestone, SpaceX 'catches' megarocket booster after test flight
-
In a first, SpaceX 'catches' megarocket booster after test flight

-
 Bangladeshi Hindus shrug off attack worries to celebrate festival
Bangladeshi Hindus shrug off attack worries to celebrate festival
-
Ubisoft fears assassin's hit over falling sales

-
 Vietnam, China hold talks on calming South China Sea tensions
Vietnam, China hold talks on calming South China Sea tensions
-
SpaceX will try to 'catch' giant Starship rocket shortly before landing

-
 Japan's former empress Michiko discharged after surgery: reports
Japan's former empress Michiko discharged after surgery: reports
-
Japan's former empress Michiko discharged after surgey: reports

-
 'Little Gregory' murder haunts France 40 years on
'Little Gregory' murder haunts France 40 years on
-
Tariffs, tax cuts, energy: What is in Trump's economic plan?

-
 Amazon wants to be everything to everyone
Amazon wants to be everything to everyone
-
Jewish school in Canada hit by gunfire for second time

-
 With medical report Harris seeks to play health card against Trump
With medical report Harris seeks to play health card against Trump
-
China-EU EV tariff talks in Brussels end with 'major differences': Beijing

-
 Buried Nazi past haunts Athens on liberation anniversary
Buried Nazi past haunts Athens on liberation anniversary
-
Harris to release medical report confirming fitness for presidency: campaign

-
 Nobel prize a timely reminder, Hiroshima locals say
Nobel prize a timely reminder, Hiroshima locals say
-
China offers $325 bn in fiscal stimulus for ailing economy

-
 Small Quebec company dominates one part of NHL hockey: jerseys
Small Quebec company dominates one part of NHL hockey: jerseys
-
Boeing to cut 10% of workforce as it sees big Q3 loss

-
 Want to film in Paris? No sexism allowed
Want to film in Paris? No sexism allowed
-
US, European markets rise as investors weigh rates, earnings

-
 In Colombia, children trade plastic waste for school supplies
In Colombia, children trade plastic waste for school supplies
-
JPMorgan Chase profits top estimates, bank sees 'resilient' US economy

-
 Little progress at key meet ahead of COP29 climate summit
Little progress at key meet ahead of COP29 climate summit
-
'Party atmosphere': Skygazers treated to another aurora show

-
 Kyrgyzstan opens rare probe into glacier destruction
Kyrgyzstan opens rare probe into glacier destruction
-
European Mediterranean states discuss Middle East, migration

-
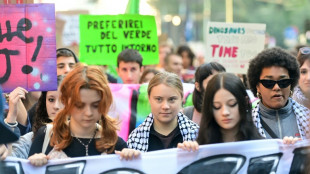 Thunberg leads pro-Palestinian, climate protest in Milan
Thunberg leads pro-Palestinian, climate protest in Milan
-
Stock markets diverge before China weekend briefing

-
 EU questions shopping app Temu over illegal products risk
EU questions shopping app Temu over illegal products risk
-
Han Kang's books sell out in South Korea after Nobel win

-
 Shanghai markets sink ahead of briefing on mixed day for Asia
Shanghai markets sink ahead of briefing on mixed day for Asia
-
Investors, analysts eye bigger China stimulus at Saturday briefing

-
 Musk unveils robotaxi, pledges it 'before 2027'
Musk unveils robotaxi, pledges it 'before 2027'
-
At least 11 dead in Florida but Hurricane Milton not as bad as feared

-
 Asian markets mixed after Wall St drop, Shanghai dips before briefing
Asian markets mixed after Wall St drop, Shanghai dips before briefing
-
Automaker Stellantis says CEO will retire in 2026


Positive results for potential first chikungunya vaccine
French-Austrian drugmaker Valneva's vaccine candidate against chikungunya, a mosquito-borne virus that causes occasional outbreaks across the world, showed promising results in a large new trial, a study said on Tuesday.
While the results were hailed as good news in the fight against chikungunya, the trial was carried out on people in the United States, where the virus is extremely rare, with experts saying more research was needed.
There are currently no vaccines or treatments available for the virus, which causes fever and sometimes debilitating joint pain, though it is rarely fatal.
Valneva says that its vaccine candidate, called VLA1553, is the first to be reviewed by health authorities after applying for approval in the US and Canada.
The new randomised, placebo-controlled phase three trial aimed to find out how often the live-attenuated vaccine, which uses a weakened form of the virus, produced an immune response.
Out of a subgroup of 266 people who received the vaccine, 263 -- 99 percent -- developed antibodies that could neutralise the chikungunya virus, according to the study published in The Lancet journal.
In a broader trial of 4,100 healthy adults, the single-shot vaccine was deemed "generally safe", with side effects similar to other vaccines.
Only two people developed serious side effects linked to the vaccine, the study said, and both fully recovered.
Martina Schneider, Valneva's clinical strategy manager and the lead author of the study, called the results "promising".
"This could be the first chikungunya vaccine available for people living in endemic regions, as well as for travellers to endemic areas or areas at risk for an upcoming outbreak," she said in a statement.
- Outbreak trials 'crucial' -
Public health experts have expressed concerns that chikungunya could be a potential future pandemic threat as climate change pushes the mosquitoes that spread it into new regions.
Kathryn Stephenson, an infectious disease specialist at the Beth Israel Deaconess Medical Center in the US, said the new study was "good news for chikungunya virus pandemic preparedness".
But the vaccine might be less effective in areas with a built-up immunity to chikungunya, which can happen with such live-attenuated shots, she added.
Since chikungunya was first identified in Tanzania in 1952, it has been recorded in more than 110 countries, according to the World Health Organization.
Occasional but severe outbreaks take place in Africa, Asia and the Americas.
Valneva has said the US Food and Drug Administration could make a decision on approving the vaccine at the end of August.
The biotech firm told AFP that it plans to submit an application to the European Union's drug watchdog, the EMA, in the second half of 2023.
The company is also trialling the vaccine among adolescents in areas of Brazil where the virus is endemic.
Stephenson said the Brazilian trial and further research conducted during actual chikungunya outbreaks were "crucial" to establishing the vaccine's effectiveness.
A chikungunya vaccine candidate developed by Denmark's Bavarian Nordic is also undergoing phase three trials.
D.Goldberg--CPN
